Projects
Our Research
We are interested in understanding how the cardiac endothelium regulates different aspects of cardiac regeneration and development and how alterations in the coronary network formation impact the ability of coronary vessels to support tissue replenishment. We use zebrafish genetic models, molecular biology, histology, in vitro mammalian models and all that with a good pinch of cool imaging.
Angiocrines in heart regeneration
Endothelial cells are the most abundant cell type in the heart. Either the ones in the endocardium or building the coronary vasculature, endothelial cells are active components of the heart with known critical roles during homeostasis and pathology. Classically, the endothelium has been perceived as a key structural component of the circulatory network. However, endothelial cells play a much more pro-active role in tissue biology. The endothelium can signal neighbouring cells by producing factors termed angiocrines. Angiocrine factors have increasingly gained attention due to their ability to regulate a broad range of processes. We and others have shown that the cardiac endothelium produces angiocrines after injury and that these factors are key to tissue repair and regeneration. Due to the nature and capabilities of angiocrines, our lab is interested in identifying pro-regenerative cardiac angiocrines and determining their mode of action.
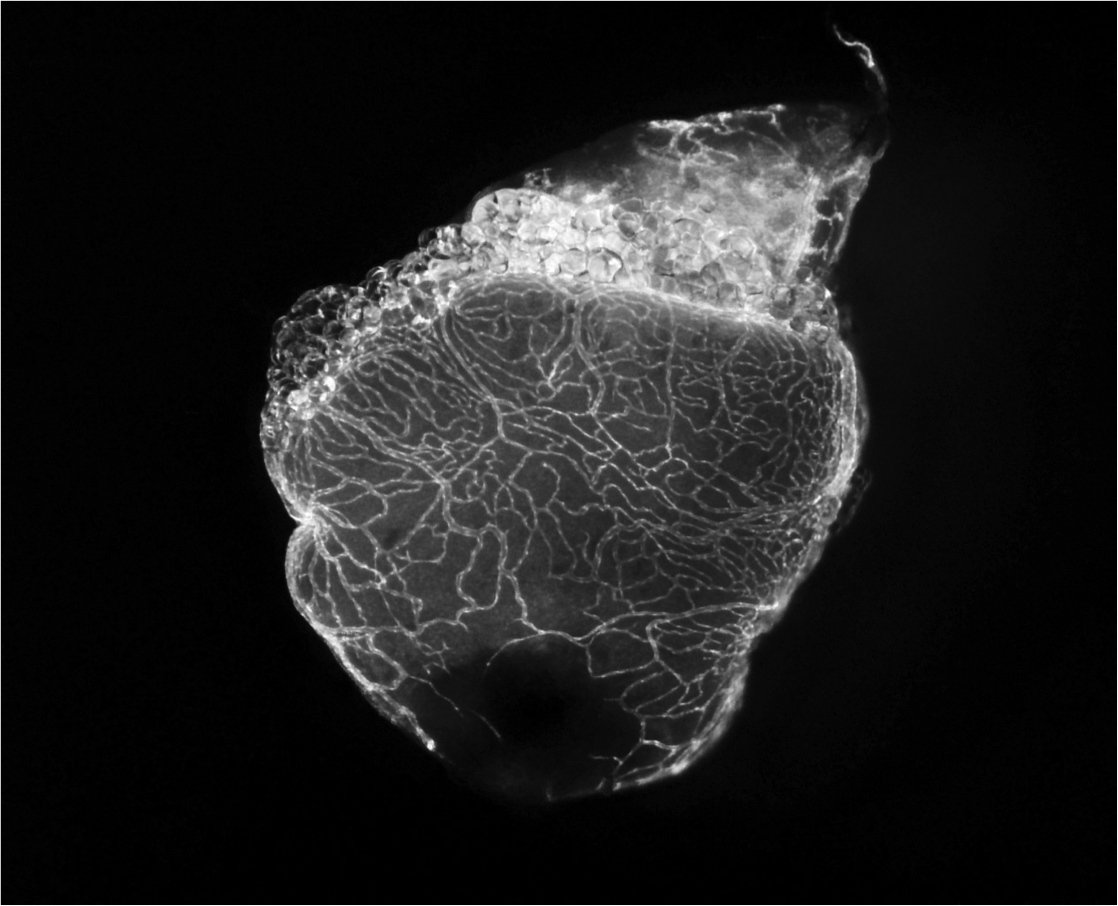
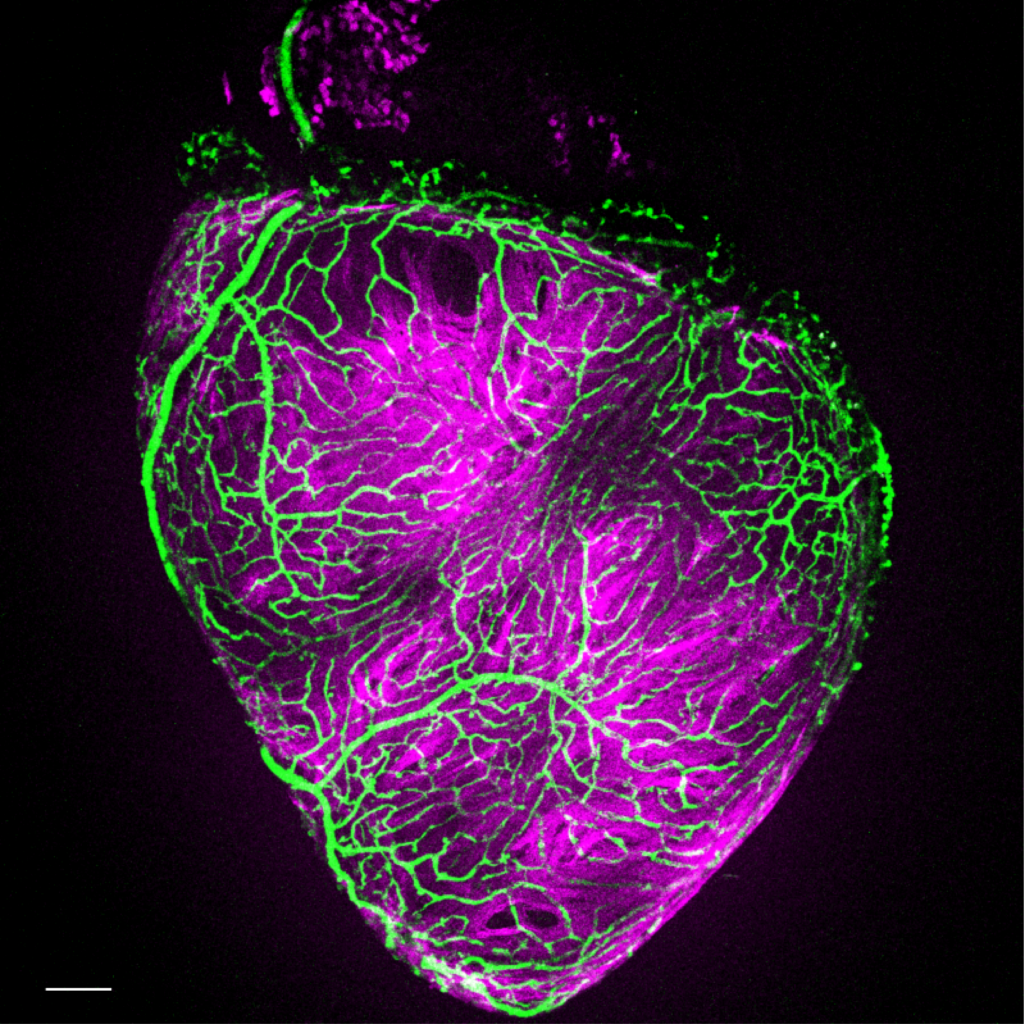
Revascularization after cardiac damage
Cardiac muscle re-perfusion is essential after myocardial infarction. Humans have a very limited capacity to efficiently grow new vessels (collaterals) to irrigate the damaged portion of the ventricle. On the contrary, zebrafish coronary vessels respond to injury by rapidly proliferating and growing, building a vascular network covering the injured area in few days. This response is critical to heart regeneration and limited to early stages after injury. Recapitulating mechanisms regulating the efficient revascularization of the zebrafish heart might have important therapeutic implications. We are interested in identifying important regulators of vascular biology involved in different aspects of revascularization and angiogenesis including sprouting, migration, specification and maturation.
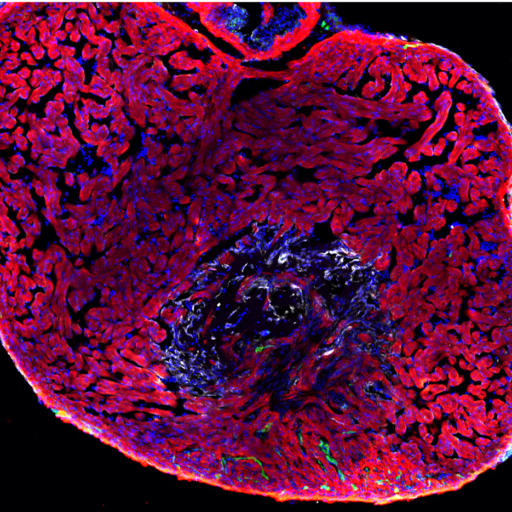
Coronaries and the developing heart
Coronary network growth and maturation are essential to cardiac development. Developmental features are recapitulated during cardiac regeneration, where damaged tissues reactivate embryonic programs to fulfill the more dynamic demands that a regenerating tissue is subjected to. Building on our expertise in tissue regeneration, we investigate how the heart develops from an endothelial-centric perspective. We found that developing vessels might be playing roles besides irrigating the muscle. Taking advantage of our privileged location in one of the biggest Pediatric hospitals of North America, our state-of-the-art imaging facilities and our beloved zebrafish, we are interested in understanding how coronary vessels interact and signal other tissues during heart development and how alterations in this crosstalk could lead to pathology.
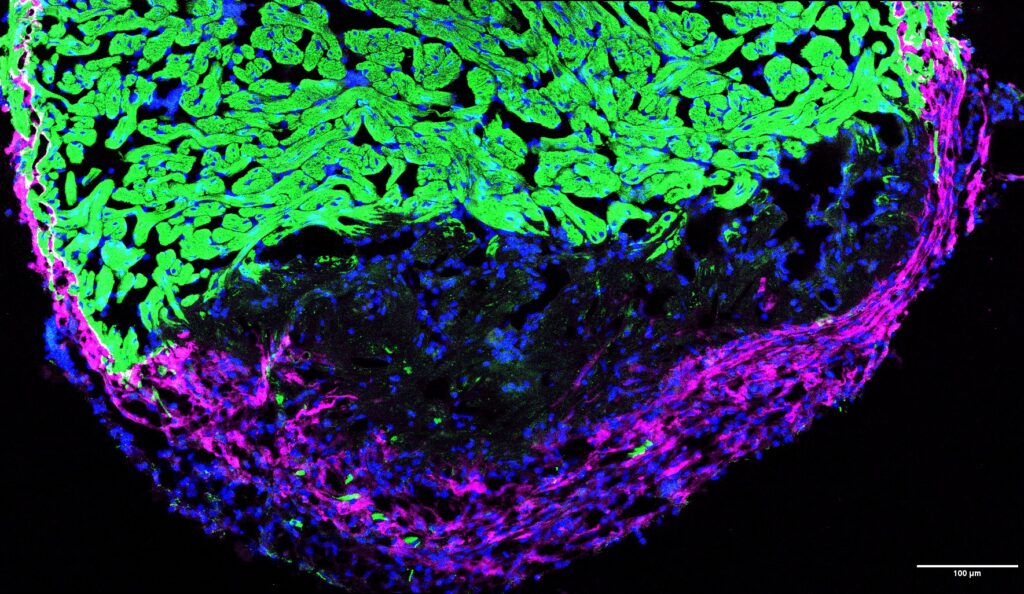
ECM/Fibrosis in repair and regeneration
In adult mammals, cardiac injury triggers a persistent fibrotic response that results in a permanent collagenous scar. While this reparative fibrosis prevents immediate heart rupture, it ultimately leads to stiffening, loss of contractility, and heart failure. Interestingly, this response is transient in the regenerative zebrafish heart.
We investigate the molecular and cellular dynamics of, treating the fibrotic niche as a transient, pro-regenerative environment rather than a terminal barrier. Our work seeks to decode the signaling networks that prevent the transition from temporary matrix remodeling to chronic scarring. By understanding how regenerative species orchestrate this balance, we aim to develop innovative strategies to “reprogram” the fibrotic response in humans, transforming permanent repair into true functional regeneration.
Immune regulation of cardiac regeneration
In species capable of complete heart regeneration, like the zebrafish, the immune response is a tightly coordinated signaling hub that instructs tissue reconstitution.
Our work focuses on the dynamic interplay between innate immune cells and the damaged tissues (e.g., myocardium, endothelium). We investigate how these cells transition from a pro-inflammatory state to a pro-regenerative phenotype, secreting specific factors that trigger tissue replenishment and prevent scarring. Our laboratory seeks to identify the specific molecular cues that allow some species to resolve inflammation without progressing to heart failure.